Amino acid sensor conserved from bacteria to humans
Abstract
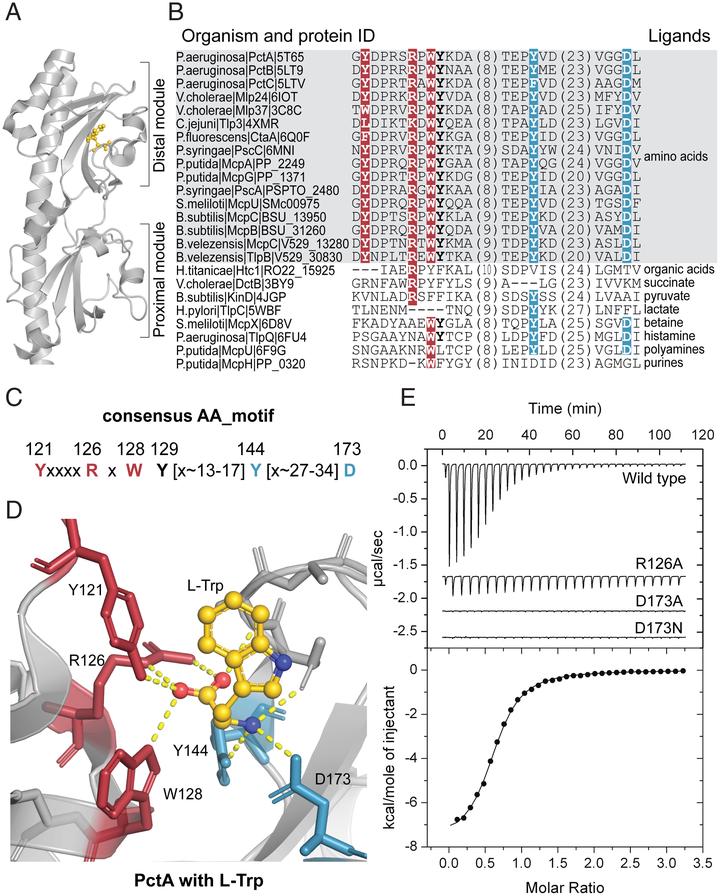
Amino acids are the building blocks of life, and they are also recognized as signals by various receptors in bacteria, archaea, and eukaryotes. Despite their common basic structure, no universal mechanism for amino acid recognition is currently known. Here, we show that a subclass of dCache_1 (double domain found in calcium channels and chemotaxis receptors, family 1), a ubiquitous extracellular sensory domain, contains a simple motif, which recognizes the amino and carboxyl groups of amino acid ligands. We found this motif throughout the Tree of Life. In bacteria and archaea, this motif exclusively binds amino acids, including γ-aminobutyric acid (GABA), and it is present in all major receptor types. In humans, this motif is found in α2δ-subunits of voltage-gated calcium channels that are implicated in neuropathic pain and neurodevelopmental disorders and in a recently characterized CACHD1 protein. Our findings suggest that GABA-derived drugs bind to the same motif in human α2δ-subunits that binds natural GABA ligands in bacterial chemoreceptors. The exact location on the target protein and the mechanism of binding may enable future improvements of drugs targeting pain and neurobiological disorders.